For most consumers, a skincare routine is a positive part of their daily routine. Thirty-eight percent (38%) see it as ‘me’ time. Sixty-three percent (63%) wash their face at least two times per day,1 yet roughly half of consumers (men and women) report using bath wash or hand soap to cleanse their face.2
So it’s no surprise that over half of consumers surveyed have no idea what ingredients to look for when purchasing a facial cleanser or understand the purpose of those ingredients. But when it comes to the aesthetic experience – 82% prefer a foaming cleanser and believe foaming is more effective than non-foaming cleansing.3
What are surfactants?
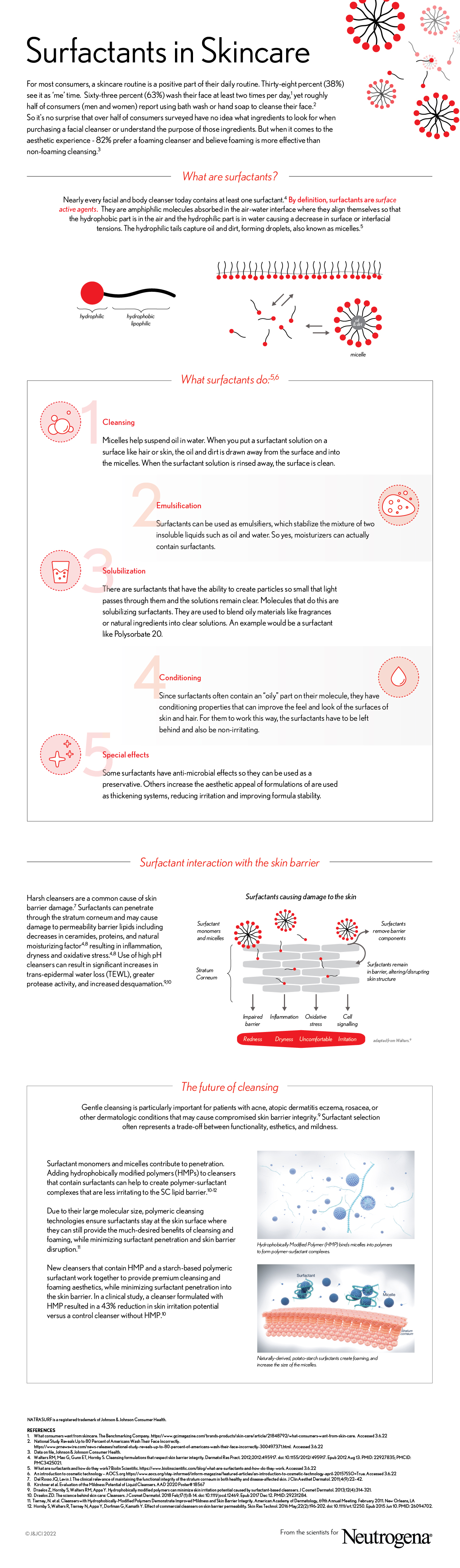
Nearly every facial and body cleanser today contains at least one surfactant.4 By definition, surfactants are surface active agents. They are amphiphilic molecules absorbed in the air-water interface where they align themselves so that the hydrophobic part is in the air and the hydrophilic part is in water causing a decrease in surface or interfacial tensions. The hydrophilic tails capture oil and dirt, forming droplets, also known as micelles.5
What surfactants do:4,5
-
- Cleansing
Micelles help suspend oil in water. When you put a surfactant solution on a surface like hair or skin, the oil and dirt is drawn away from the surface and into the micelles. When the surfactant solution is rinsed away, the surface is clean.
-
- Emulsification
Surfactants can be used as emulsifiers, which stabilize the mixture of two insoluble liquids such as oil and water. So yes, moisturizers can actually contain surfactants.
-
- Solubilization
There are surfactants that have the ability to create particles so small that light passes through them and the solutions remain clear. Molecules that do this are solubilizing surfactants. They are used to blend oily materials like fragrances or natural ingredients into clear solutions. An example would be a surfactant like Polysorbate 20.
-
- Conditioning
Since surfactants often contain an “oily” part on their molecule, they have conditioning properties that can improve the feel and look of the surfaces of skin and hair. For them to work this way, the surfactants have to be left behind and also be non-irritating.
-
- Special effects
Some surfactants have anti-microbial effects so they can be used as a preservative. Others increase the aesthetic appeal of formulations of are used as thickening systems, reducing irritation and improving formula stability.
Surfactant Interaction with the Skin Barrier
Harsh cleansers are a common cause of skin barrier damage.7 Surfactants can penetrate through the stratum corneum and may cause damage to permeability barrier lipids including decreases in ceramides, proteins, and natural moisturizing factor4,8 resulting in inflammation, dryness and oxidative stress.4,8 Use of high pH cleansers can result in significant increases in trans-epidermal water loss (TEWL), greater protease activity, and increased desquamation.9,10
The Future of Cleansing
Gentle cleansing is particularly important for patients with acne, atopic dermatitis eczema, rosacea, or other dermatologic conditions that may cause compromised skin barrier integrity.9 Surfactant selection often represents a trade-off between functionality, esthetics, and mildness. Surfactant monomers and micelles contribute to penetration. Adding hydrophobically modified polymers (HMPs) to cleansers that contain surfactants can help to create polymer-surfactant complexes that are less irritating to the SC lipid barrier.10-12
Due to their large molecular size, polymeric cleansing technologies ensure surfactants stay at the skin surface where they can still provide the much-desired benefits of cleansing and foaming, while minimizing surfactant penetration and skin barrier disruption.11
New cleansers that contain HMP and a starch-based polymeric surfactant work together to provide premium cleansing and foaming aesthetics, while minimizing surfactant penetration into the skin barrier. In a clinical study, a cleanser formulated with HMP resulted in a 43% reduction in skin irritation potential versus a control cleanser without HMP.10
To learn more about surfactants in skincare, see the infographic below.
CLICK ON IMAGE TO ENLARGE AND DOWNLOAD
For additional resources and products that can help you and your patients, visit NeutrogenaMD.com
NATRASURF is a registered trademark of Johnson & Johnson Consumer Health.
REFERENCES
-
- What consumers want from skincare. The Benchmarking Company. https://www.gcimagazine.com/brands-products/skin-care/article/21848792/what-consumers-want-from-skin-care. Accessed 3.6.22
- National Study Reveals Up to 80 Percent of Americans Wash Their Face Incorrectly. https://www.prnewswire.com/news-releases/national-study-reveals-up-to-80-percent-of-americans-wash-their-face-incorrectly-300497371.html. Accessed 3.6.22
- Data on file, Johnson & Johnson Consumer Health.
- Walters RM, Mao G, Gunn ET, Hornby S. Cleansing formulations that respect skin barrier integrity. Dermatol Res Pract. 2012;2012:495917. doi: 10.1155/2012/495917. Epub 2012 Aug 13. PMID: 22927835; PMCID: PMC3425021.
- What are surfactants and how do they work? Biolin Scientific. https://www.biolinscientific.com/blog/what-are-surfactants-and-how-do-they-work. Accessed 3.6.22
- An introduction to cosmetic technology – AOCS.org https://www.aocs.org/stay-informed/inform-magazine/featured-articles/an-introduction-to-cosmetic-technology-april-2015?SSO=True. Accessed 3.6.22
- Del Rosso JQ, Levin J. The clinical relevance of maintaining the functional integrity of the stratum corneum in both healthy and disease-affected skin. J Clin Aesthet Dermatol. 2011;4(9):22–42.
- Kirchner et al. Evaluation of the Mildness Potential of Liquid Cleansers. AAD 2020 Poster# 18567
- Draelos Z, Hornby S, Walters RM, Appa Y. Hydrophobically modified polymers can minimize skin irritation potential caused by surfactant-based cleansers. J Cosmet Dermatol. 2013;12(4):314-321.
- Draelos ZD. The science behind skin care: Cleansers. J Cosmet Dermatol. 2018 Feb;17(1):8-14. doi: 10.1111/jocd.12469. Epub 2017 Dec 12. PMID: 29231284.
- Tierney, N. et al. Cleansers with Hydrophobically-Modified Polymers Demonstrate Improved Mildness and Skin Barrier Integrity. American Academy of Dermatology, 69th Annual Meeting. February 2011. New Orleans, LA
- Hornby S, Walters R, Tierney N, Appa Y, Dorfman G, Kamath Y. Effect of commercial cleansers on skin barrier permeability. Skin Res Technol. 2016 May;22(2):196-202. doi: 10.1111/srt.12250. Epub 2015 Jun 10. PMID: 26094702.
Did you enjoy this article? You can find more on our OTC Resource Center.