CASE
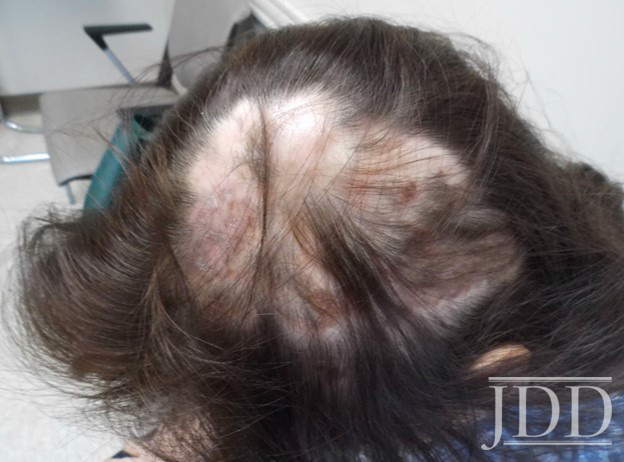
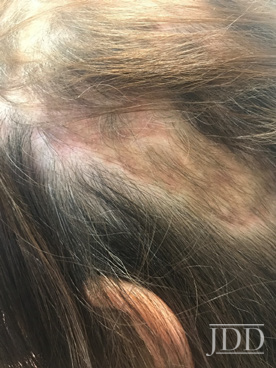
A 48-year-old female with a history of systemic lupus erythematosus (SLE) treated with hydroxychloroquine and discoid lupus erythematosus (DLE) presented with several dyspigmented plaques and significant scarring alopecia of the scalp. The patient experienced improvement in her hair loss initially and was stable using topical corticosteroids and subsequently intralesional Triamcinolone injections. After being lost to follow up for over a year, she reverted to worsening of her DLE with increased areas of scarring alopecia. Examination at that time revealed several smooth, hyperpigmented, atrophic and irregular patches of alopecia more significant on the left than right parietal scalp and on the frontal scalp. A few areas showed decreased follicles with overlying adherent scale on the hyperpigmented patches (Figure 1). The patient, co-managed by rheumatology, declined any additional systemic medications for her hair loss and flaring DLE due to other health co-morbidities (previous history of breast carcinoma). Although rheumatology recommended treatment with belimumab, the patient was unable to tolerate this medication in the past and instead elected to try PRP.
22 mL of the patient’s blood was obtained using the Eclipse PRP patient kit. The PRP was prepared using the standardized Eclipse protocol (Eclipse Aesthetics, LLC, Dallas, TX). The blood was centrifuged at 3500 revolutions per minute for 10 minutes and yielded a red blood cell precipitate and plasma supernatant. The platelet poor plasma or upper portion of the supernatant was then removed while the PRP portion of the blood remained for use. After cleaning and prepping the site, the patient’s areas of scarring alopecia on the scalp were injected subcutaneously using a 30-gauge needle with a total volume of approximately 7 mL of PRP.
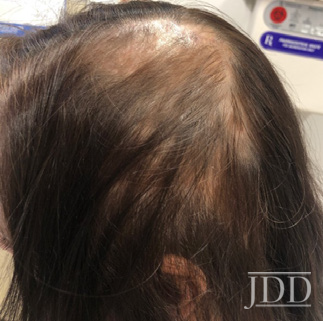
After three treatment sessions of PRP injections (spaced 3 months apart given patient scheduling constraints), significant regrowth was noted overall via global photography, with improvement seen even within the areas of scarring alopecia (Figure 2).
DISCUSSION
While several studies have shown PRP’s success in treating nonscarring hair loss, few documented reports of cicatricial alopecia treated with PRP are found in the literature to date. One is a two patient case series of PRP use in centrifugal cicatricial alopecia (CCCA) and lichen planopilaris (LPP)1; there are two additional case reports of PRP used to treat LPP4 and one case report of its use in frontal fibrosing alopecia (FFA).5
Published data emphasizes PRP’s ability to improve the number of hair follicles and bulge cells, vascularity, and epidermal thickness.6 More specifically, platelet activation releases proteins and cytokines that induce folliculogenesis, promote the anagen growing phase of dormant bulbs, and are antiinflammatory. 1,3 While these effects may play a role in scarring alopecia, additional studies are needed to further elucidate the mechanism and efficacy of PRP in these patients.
In summary, the authors’ case is one of very few examples that suggest clinical benefit with the use of PRP as a treatment for cicatricial alopecia and the only case of PRP used for DLE-associated scarring alopecia reported in the literature. The exact mechanism for this promising treatment remains uncertain and additional controlled trials are needed to better understand the role of PRP in both cicatricial and non-cicatricial alopecia.
DISCLOSURES
REFERENCES
SOURCE
Polster, H., Kagha, K., & Luke, J. (2022). Platelet Rich Plasma for the Treatment of Scarring Alopecia Due to Discoid Lupus Erythematosus. Journal of Drugs in Dermatology: JDD, 21(3), 309-310.
Content and images used with permission from the Journal of Drugs in Dermatology.
Adapted from original article for length and style.